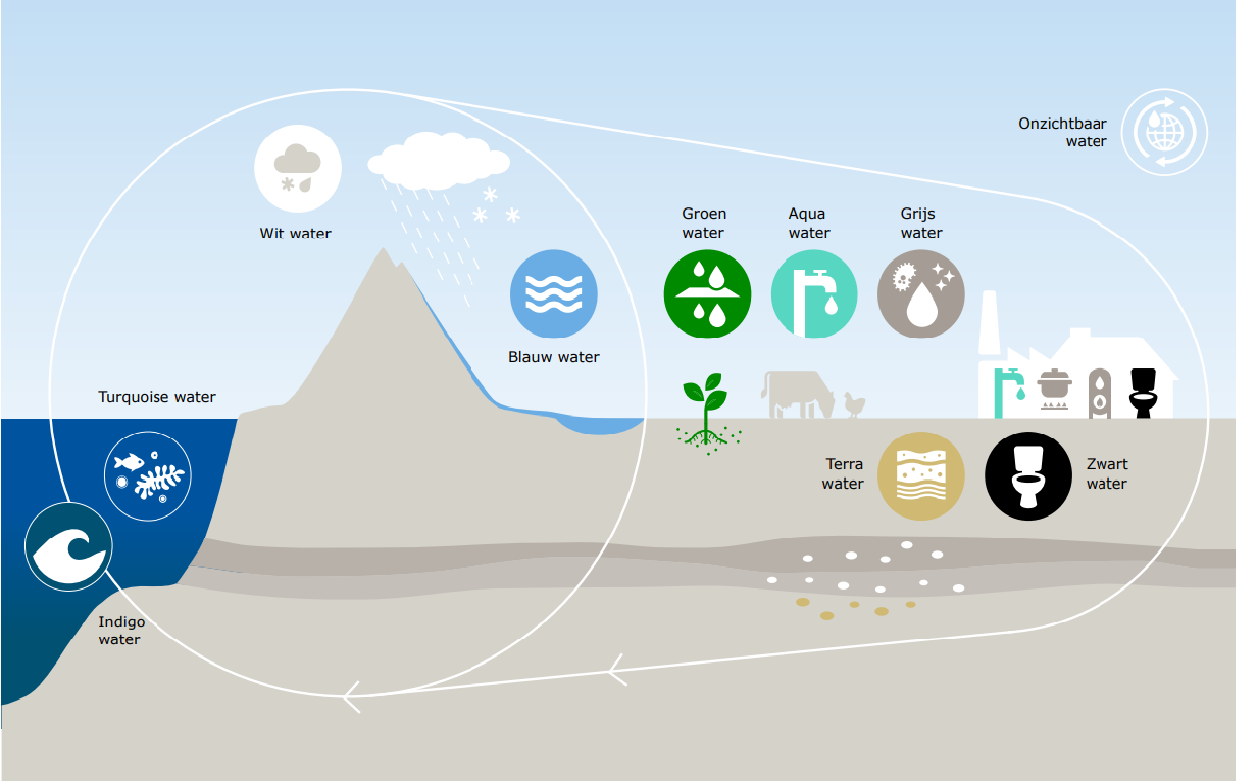
Producing sweet water from saltwater is becoming increasingly relevant, considering the growing shortage of water. Membranes are frequently used to desalinate water. However, seawater also contains pollutants such as boron ions, which is toxic in high concentrations.
Boron and some other ions are difficult to extract from water due to their chemical properties. They are amphoteric: the PH value influences their properties. ‘The standard membrane technology is insufficient to extract these particles from the water’, says Jouke Dykstra, associate professor of Environmental Technology. ‘Chemicals must be added to adjust the PH value, which is something we want to avoid.’
Model
In collaboration with scientists from the Technion research institute in Israel and Wetsus in Leeuwarden, Dykstra developed a new theoretical model. This enables researchers to predict the behaviour of the boron ions in the water treatment. This, in turn, allowed them to design a process for the removal of boron during desalination. To this end, they deploy the capacitive deionisation method, a new, membrane-less method through which the water flows through carbon electrodes with micro-pores. The electric current causes the ions to adsorb to the electrodes and thus be removed from the water. The study was published in PNAS.
Dykstra: ‘We are the first to develop a model to predict this behaviour. This enables us to steer chemical processes.’ Dykstra states that this model may also be used for other water-related issues, such as removing arsenic, medicine residues or herbicides.’

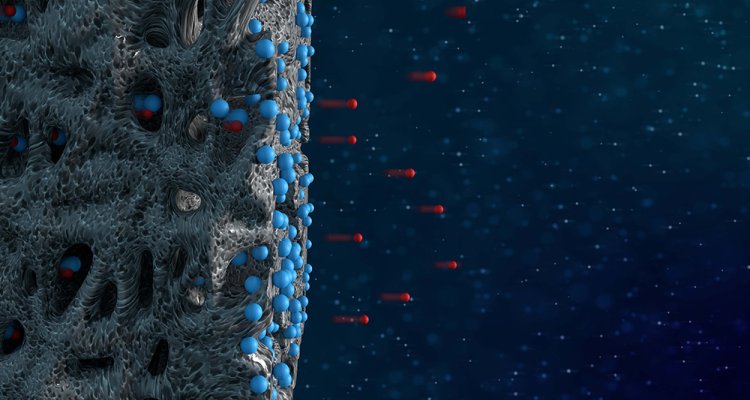 Photo: Electrodes absorb pollutants in saltwater. Illustration Paul Gerlach
Photo: Electrodes absorb pollutants in saltwater. Illustration Paul Gerlach